
"Stability Without Membranes: 'Dome Device' Technique for Larger Bone Defects"
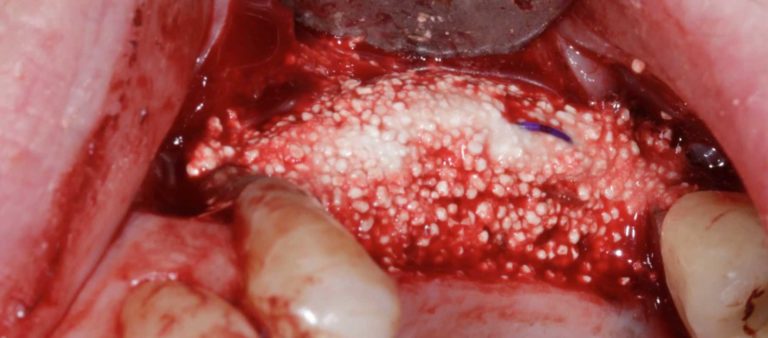
In this webinar, Dr Mike Ainsworth discusses this exciting technique, utilising EthOss bone graft material and a “dome device” to create a stable graft, without a collagen membrane, even in larger defects.
Created using short lengths of resorbable sutures, the dome device helps to reliably prevent soft tissue pressure from disturbing the graft, whilst still being fully replaced by patient host bone.
The discussion includes:
- • Overview of published research outlining the “dome device” technique and its potential to increase stability
- • Clinical challenges (and potential solutions) in creating the “dome device”
- • Case studies discussing utilising the technique in practice
Dr Ainsworth published in the European Journal for Dental Implantologists (Issue 1/19) demonstrating this technique – the paper can be viewed here.
About the speaker:
Michael Ainsworth graduated from Birmingham University in 2002 and has been in private practice since that time. He became very interested in complex and cosmetic dentistry immediately, attending many courses and conferences both at home and abroad and started placing implants under supervision soon thereafter, completing a 1-year certification course in 2004.
Over the course of the last decade and under the mentorship of Peter Fairbairn he has developed a keen interest in streamlining techniques for the use of synthetic biomaterials in bone grafting, and soft tissue development.
In 2007 he moved to Spain to run a private practice. Since returning to the UK he has worked in a number of practices in Yorkshire and the West Midlands. He is currently undertaking an MSc in Periodontology and has limited his practice to dental implant treatments.
Watch now
Enter your details below to access this webinar
Disclaimer
Ethoss Regeneration Ltd (“Us”, “Our”) will not be liable, or accept responsibility for, any use or application of the products, or the techniques, mentioned in any case studies, articles, newsletters, blogs, brochures or any other media prepared by Us or contained in Our literature, website, social media channels or any other communications. It is the clinician’s responsibility to follow the official guidance related to each of the products as produced by the relevant manufacturer and ensure they are adhering to recommended methods and uses and complying with the correct techniques for use of the products.
All case studies and clinical examples are for illustrative purposes only. Please always refer to the product manufacturer’s guidance as to how to use any products and techniques mentioned in these case studies
Join our exclusive On-demand Training Academy
Gain access to 6 video training modules and advance through the tutorials to get a simple grasp on using EthOss in various procedures.

Routine Implant Procedures - Part 1
Module 1, part 1: Dr Peter Fairbairn, 8 minutes
Dr Fairbairn discusses using EthOss to treat routine bone loss cases according to his published Protocol.
Routine Implant Procedures - Part 2
Module 1, part 2: Dr Peter Fairbairn, 12 minutes
Dr Fairbairn also discusses flap design, suture techniques and the importance of thorough cleaning / degranulation prior to grafting.
Socket Grafting
Module 2: Dr Peter Fairbairn, 15 minutes
Advice and techniques for optimising socket grafting outcomes with EthOss, includes options for covering the site and how to effectively clean extraction sockets prior to graft placement
